Q 1 – A concave mirror forms a real image twice the size of the object. If the object distance is 15 cm, the focal length is:
(a) –10 cm
(b) –15 cm
(c) –30 cm
(d) –7.5 cm
(d) –7.5 cm
Q 2 – The radius of curvature of a mirror is +40 cm. The nature of mirror is:
(a) Concave
(b) Convex
(c) Plane
(d) Cannot be determined
(b) Convex
Q 3 – An object is placed at the centre of curvature of a concave mirror. The image formed will be:
(a) Virtual, erect, same size
(b) Real, inverted, same size
(c) Real, inverted, magnified
(d) Virtual, erect, diminished
(b) Real, inverted, same size
Q 4 – Which mirror is used as rear-view mirror in vehicles and why?
(a) Concave, large image
(b) Convex, wider field of view
(c) Plane, erect image
(d) Concave, real image
(b) Convex, wider field of view
Q 5 – A ray parallel to principal axis after reflection passes through focus. This applies to:
(a) Convex mirror
(b) Concave mirror
(c) Plane mirror
(d) Both (a) and (c)
(b) Concave mirror
Q 6 – Which condition will stop photosynthesis completely and immediately?
(a) Removal of nitrogen
(b) Absence of water
(c) Absence of light
(d) Lack of minerals
(c) Absence of light
Q 7 – In a destarched plant, iodine test after exposure to light shows starch only in green parts because:
(a) Only green parts absorb water
(b) Chlorophyll is required for photosynthesis
(c) Only green parts respire
(d) Only green parts have stomata
(b) Chlorophyll is required for photosynthesis
Q 8 – Which of the following is NOT a function of stomata?
(a) Gas exchange
(b) Transpiration
(c) Food synthesis
(d) Regulation of water
(c) Food synthesis
Q 9 – In Amoeba, ingestion of food occurs by:
(a) Diffusion
(b) Endocytosis
(c) Active transport
(d) Osmosis
(b) Endocytosis
Q 10 – Digestion in Amoeba is:
(a) Only extracellular
(b) Only intracellular
(c) Both
(d) None
(b) Only intracellular
Q 11 – Which enzyme starts carbohydrate digestion in animals?
(a) Pepsin
(b) Lipase
(c) Amylase
(d) Trypsin
(c) Amylase
Q 12 – Which organ is responsible for emulsification of fats?
(a) Stomach
(b) Liver
(c) Pancreas
(d) Mouth
(b) Liver
Q 13 – The semi-digested food leaving the stomach is called:
(a) Bolus
(b) Chyme
(c) Faeces
(d) Plasma
(b) Chyme
Q 14 – Which step of nutrition involves movement of digested food into blood?
(a) Ingestion
(b) Digestion
(c) Absorption
(d) Egestion
(c) Absorption
Q 15 – Which type of reaction is:
Zn + CuSO₄ → ZnSO₄ + Cu
(a) Combination
(b) Decomposition
(c) Displacement
(d) Double displacement
(c) Displacement
Q 16 – The reaction:
CaO + H₂O → Ca(OH)₂ is:
(a) Endothermic
(b) Exothermic
(c) Neutralisation
(d) Redox
(b) Exothermic
Q 17 – Oxidation involves:
(a) Gain of electrons
(b) Loss of electrons
(c) Gain of neutrons
(d) Loss of protons
(b) Loss of electrons
Q 18 – Which of the following reactions is a double displacement reaction?
(a) Na + Cl₂ → NaCl
(b) AgNO₃ + NaCl → AgCl + NaNO₃
(c) Fe + O₂ → Fe₂O₃
(d) H₂ + O₂ → H₂O
(b) AgNO₃ + NaCl → AgCl + NaNO₃
Q 19 – Rancidity can be prevented by:
(a) Heating
(b) Adding oxygen
(c) Storing in airtight containers
(d) Adding water
(c) Storing in airtight containers
Q 20 – The black coating on silver is due to formation of:
(a) Silver oxide
(b) Silver nitrate
(c) Silver sulphide
(d) Silver chloride
(c) Silver sulphide
Q 21 – Fill in the Blanks:
1. The focal length of a plane mirror is ________.
2. The image formed by a convex mirror is always ________ and ________.
3. The relation between focal length (f) and radius of curvature (R) is ________.
4. Magnification of a mirror is given by m = ________.
5. The pole of a spherical mirror lies on the ________.
6. The site of photosynthesis in plant cell is ________.
7. The finger-like projections in small intestine are called ________.
8. In Amoeba, digestion occurs in ________.
9. The basic unit of nutrition in plants is ________ (process name).
10. The process of removal of undigested food is ________.
11. A chemical equation must be ________ to obey conservation of mass.
12. Breaking down of a compound into simpler substances is called ________.
13. Rusting of iron requires ________ and ________.
14. The reaction in which two or more substances combine is called ________
1. Infinite
2. Virtual and erect & Dimished.
3. R = 2f
4. Magnification = (– v/u)
5. Principal axis
6. Chloroplast
7. Villi
8. Food vacuole
9. Photosynthesis
10. Egestion
11. Balanced
12. Decomposition
13. Oxygen and water (moisture)
14. Combination
Directions:
(A) Both Assertion and Reason are true, and Reason explains Assertion
(B) Both true, but Reason does not explain Assertion
(C) Assertion true, Reason false
(D) Assertion false, Reason true
Q 22 – Assertion: Convex mirrors always form virtual images.
Reason: Reflected rays diverge after reflection.
(A) Both Assertion and Reason are true, and Reason explains Assertion
Q 23 – Assertion: A concave mirror can form both real and virtual images.
Reason: It depends on object position relative to focus.
(A) Both Assertion and Reason are true, and Reason explains Assertion
Q 24 – Assertion: Image formed by plane mirror is laterally inverted.
Reason: Left and right sides are interchanged.
(A) Both Assertion and Reason are true, and Reason explains Assertion
Q 25 – Assertion: A real image can be obtained on a screen.
Reason: Real image is formed by actual intersection of rays.
(A) Both Assertion and Reason are true, and Reason explains Assertion
Q 26 – Assertion: The focal length of concave mirror is positive.
Reason: Sign convention follows Cartesian system.
(D) Assertion is False, Reason is True
Q 27 – Assertion: Plants kept in dark for long show no starch.
Reason: Stored starch is used up for respiration.
(A) Both Assertion and Reason are true, and Reason explains Assertion
Q 28 – Assertion: Stomata remain closed at night.
Reason: Photosynthesis does not occur in absence of light.
(A) Both Assertion and Reason are true, and Reason explains Assertion
Q 29 – Assertion: Amoeba can digest food.
Reason: It forms food vacuole containing enzymes.
(A) Both Assertion and Reason are true, and Reason explains Assertion
Q 30 – Assertion: Saliva helps in digestion.
Reason: It contains enzymes that break down starch.
(A) Both Assertion and Reason are true, and Reason explains Assertion
Q 31 – Assertion: Digestion in animals is necessary.
Reason: Complex food cannot be directly absorbed.
(A) Both Assertion and Reason are true, and Reason explains Assertion
Q 32 – Assertion: Mass is conserved in chemical reactions.
Reason: Atoms are neither created nor destroyed.
(A) Both Assertion and Reason are true, and Reason explains Assertion
Q 33 – Assertion: Rusting of iron is an oxidation reaction.
Reason: Iron gains oxygen in the reaction.
(A) Both Assertion and Reason are true, and Reason explains Assertion
Q 34 – Assertion: All decomposition reactions are endothermic.
Reason: Energy is required to break bonds.
(A) Both Assertion and Reason are true, and Reason explains Assertion
Q 35 – Assertion: Double displacement reactions always produce a precipitate.
Reason: Exchange of ions leads to insoluble product.
(D) Assertion false, Reason true
Q 36 – Assertion: Oxidation and reduction occur together.
Reason: Loss and gain of electrons happen simultaneously.
(A) Both Assertion and Reason are true, and Reason explains Assertion
Q 37 – State the laws of reflection.
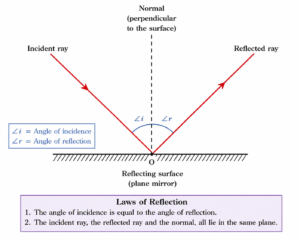
1. The angle of incidence is equal to the angle of reflection.
2. The incident ray, reflected ray, and the normal at the point of incidence lie in the same plane.
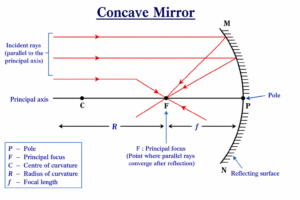
Q 38 – Define principal focus of a concave mirror.
A point on the principal axis where all rays of light parallel to the principal axis converge (meet) after reflection from the mirror.
When parallel light rays fall on a concave mirror, they reflect and come together at a single point. This point is called the principal focus (F).
Q 39 – What is magnification? Write its formula for mirrors.
Magnification is the ratio of the height of the image to the height of the object. It tells us how much bigger or smaller the image is compared to the object.

Q 40 – Why is chlorophyll essential for photosynthesis?
Chlorophyll is essential for photosynthesis because:
- It absorbs sunlight energy, mainly in the blue and red regions of the spectrum.
- This absorbed light energy is converted into chemical energy, which drives the process of making food.
- It enables the plant to convert carbon dioxide and water into glucose (food).
Q 41 – Explain why Amoeba is said to have holozoic nutrition.
Amoeba is said to have holozoic nutrition because it follows the same method of nutrition as higher animals, involving ingestion, digestion, absorption, assimilation, and egestion.
Explanation:
- Ingestion: Amoeba surrounds and engulfs food using pseudopodia (forming a food vacuole).
- Digestion: Enzymes inside the food vacuole break down the food into simpler substances.
- Absorption: The digested nutrients diffuse into the cytoplasm.
- Assimilation: The absorbed food is used for energy, growth, and repair.
- Egestion: Undigested waste is expelled out of the cell.
Q 42 – What is the role of enzymes in digestion?
Enzymes are biological catalysts that speed up the chemical breakdown of food during digestion.
Role of enzymes in digestion:
- They convert complex food substances into simpler, soluble forms that can be absorbed.
- Each enzyme is specific to a type of food:
- Amylase → breaks starch into sugars
- Protease (e.g., pepsin) → breaks proteins into amino acids
- Lipase → breaks fats into fatty acids and glycerol
- They make digestion faster and efficient at body temperature.
Q 43 – State the law of conservation of mass with example.
Law of Conservation of Mass:
The law of conservation of mass states that:
- Mass can neither be created nor destroyed in a chemical reaction.
- The total mass of reactants is equal to the total mass of products.
Example:
Consider the reaction:
2H2+O2→2H2O
- Mass of reactants:
- 2H2 = 4 g
- O2 = 32 g
- Total = 36 g
- Mass of products:
- 2H2O = 36g
Total mass before = Total mass after = 36 g
Q 44 – What is a balanced chemical equation? Why is it necessary?
A balanced chemical equation is an equation in which the number of atoms of each element is equal on both sides (reactants and products).
Example:
2H2 + O2 → 2H2
Why is it necessary?
- Follows the law of conservation of mass
- Ensures that matter is neither created nor destroyed.
- Gives correct proportions of reactants and products
- Helps in calculating how much substance is needed or formed.
- Represents the actual chemical reaction accurately
- An unbalanced equation does not show the real process.
Q 45 – Define oxidation and reduction with one example each.
Oxidation:
Oxidation is the process in which a substance gains oxygen or loses hydrogen.
Example:
2Mg+O2 → 2MgO
Here, magnesium is oxidized because it gains oxygen.
Reduction:
Reduction is the process in which a substance loses oxygen or gains hydrogen.
Example:
CuO+H2 → Cu+H2O
Here, copper oxide is reduced because it loses oxygen.
Q 46 – Explain image formation by concave mirror for the following cases with ray diagrams:
Object at infinity
Object between focus and pole
1. Object at infinity
Ray diagram idea: Parallel rays coming from a very distant object reflect and meet at the focus.
Explanation:
Rays from the object are parallel to the principal axis.
After reflection, they converge at the principal focus (F).
Image formed:
Position: At focus (F)
Nature: Real and inverted
Size: Highly diminished (point-sized)
2. Object between focus (F) and pole (P)
Ray diagram idea: Rays diverge after reflection and appear to come from behind the mirror.
Explanation:
One ray parallel to axis reflects through F.
Another ray towards pole reflects obeying laws of reflection.
Reflected rays diverge, but their backward extensions meet behind the mirror.
Image formed:
Position: Behind the mirror
Nature: Virtual and erect
Size: Magnified (enlarged)
Q 47 – An object is placed 20 cm in front of a concave mirror of focal length 10 cm. Find:
Position of image
- Nature of image
- Magnification
Answer

Q 47 – An object is placed 20 cm in front of a concave mirror of focal length 10 cm. Find:
- Position of image
- Nature of image
- Magnification
Answer

Q 48 – Explain the process of digestion in humans (mouth to small intestine). Also answer:
- Why must food be chewed properly before swallowing?
- Why does protein digestion begin only in the stomach?
- Why is the small intestine highly coiled and long?
Digestion in humans (Mouth → Small Intestine)
Mouth (Buccal cavity):
Food is taken in and chewed by teeth (mechanical digestion).
Saliva contains the enzyme salivary amylase, which begins digestion of starch into simpler sugars.
The tongue helps in mixing food and forming a bolus for swallowing.
Oesophagus (Food pipe):
Food moves to the stomach by peristalsis (wave-like muscular movement).
No digestion occurs here.
Stomach:
Food is mixed with gastric juice containing hydrochloric acid (HCl) and pepsin.
HCl makes the medium acidic and kills germs.
Pepsin begins protein digestion, converting proteins into simpler substances (peptones).
Food becomes semi-liquid called chyme.
Small Intestine:
Receives bile from liver (helps in fat digestion) and pancreatic juice.
Pancreatic enzymes digest carbohydrates, proteins, and fats completely.
Intestinal juice completes digestion into glucose, amino acids, fatty acids, and glycerol.
Q 49 – Explain the processes of absorption and assimilation in humans. Also answer:
- Why are villi important?
- What will happen if villi are damaged or absent?
- Why is absorption maximum in the small intestine?
Absorption and Assimilation in Humans
Absorption:
Absorption is the process by which digested food (glucose, amino acids, fatty acids, glycerol, etc.) passes through the walls of the small intestine into the blood or lymph.
It mainly occurs in the small intestine through tiny finger-like projections called villi.
Assimilation:
Assimilation is the process by which the absorbed nutrients are transported to body cells and used for energy, growth, repair, and storage.
For example, glucose is used in respiration, proteins help in growth and repair.
1. Why are villi important?
- Villi increase the surface area for absorption.
- They contain blood capillaries and lacteals, which help in quick transport of nutrients.
2. What will happen if villi are damaged or absent?
- Absorption of nutrients will be poor or incomplete.
- This can lead to malnutrition, (weakness), and deficiency diseases.
3. Why is absorption maximum in the small intestine?
- It is long and highly coiled, providing more surface area.
- Presence of villi and microvilli increases absorption efficiency.
- Rich blood supply helps rapid transport of nutrients.
Q 50 – Explain the role of digestive enzymes in human digestion. Include:
- Enzymes in mouth, stomach, and small intestine
- Their specific functions
- What will happen if enzymes are absent or inactive
Digestive enzymes are biological catalysts that break down complex food into simpler, absorbable forms.
• Mouth: Salivary amylase converts starch into simple sugars (maltose).
• Stomach: Pepsin breaks proteins into peptides in an acidic medium.
• Small intestine:
– Trypsin further digests proteins into amino acids
– Amylase continues carbohydrate digestion
– Lipase breaks fats into fatty acids and glycerol
If enzymes are absent or inactive:
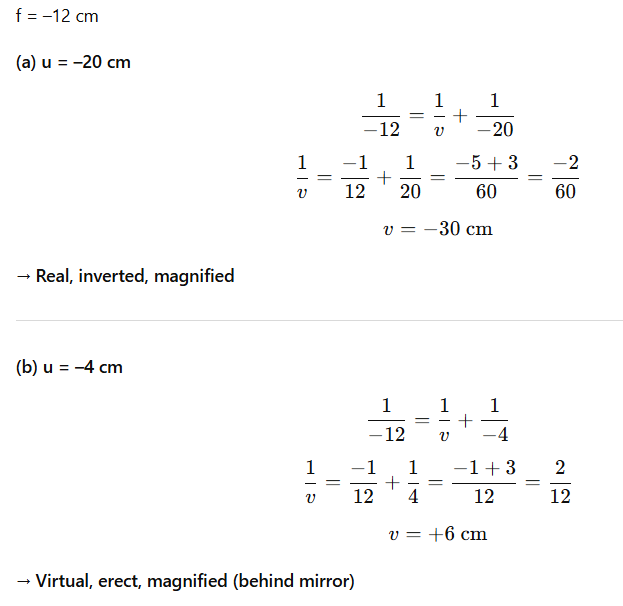
Food will remain undigested, leading to poor absorption, malnutrition, and digestive disorders.
Q 51 – Balance the following equation and identify the type of reaction:
Fe + H₂O → Fe₃O₄ + H₂
Also explain the steps used in balancing.
Answer:
Type of Reaction:
This is a redox reaction because:
- Iron (Fe) is oxidized to Fe3O4(gains oxygen)
- Hydrogen (H₂O) is reduced to H2 (loses oxygen)
(It can also be seen as a displacement reaction, where iron displaces hydrogen from water.)

- Balanced equation:
- Fe + H₂O → Fe₃O₄ + H₂
- Type: Redox reaction (also displacement reaction)
Q 52 – An object 20 cm from a spherical mirror gives rise to a virtual image 15 cm behind the mirror. Determine the magnification of the image and the type of mirror used.

Q 53 – At what distance from a concave mirror of focal length 10 cm should an object be placed so that
a) its real image is formed 20 cm from the mirror?
b) its virtual image is formed 20 cm from the mirror?

Q 54 – An object is placed (a) 20 cm, (b) 4 cm, in front of a concave mirror of focal length 12 cm. Find the nature and position of the image formed in each case.